Live Pain Free with The
Discseel® Procedure
Your active lifestyle doesn’t have to stop because you suffer from Degenerative Disc Disease. There is hope for relief from chronic back pain with the Discseel Procedure.
The Discseel® Procedure's Success
Hear what our past patients have to say about the Discseel Procedure
- of Discseel® patients report an improvement and a reduction in overall pain
- of patients who previously had a failed spinal surgery get relief from a Spinal Fusion
- Statistic is measured across all locations in the United States




What Is The Discseel® Procedure?
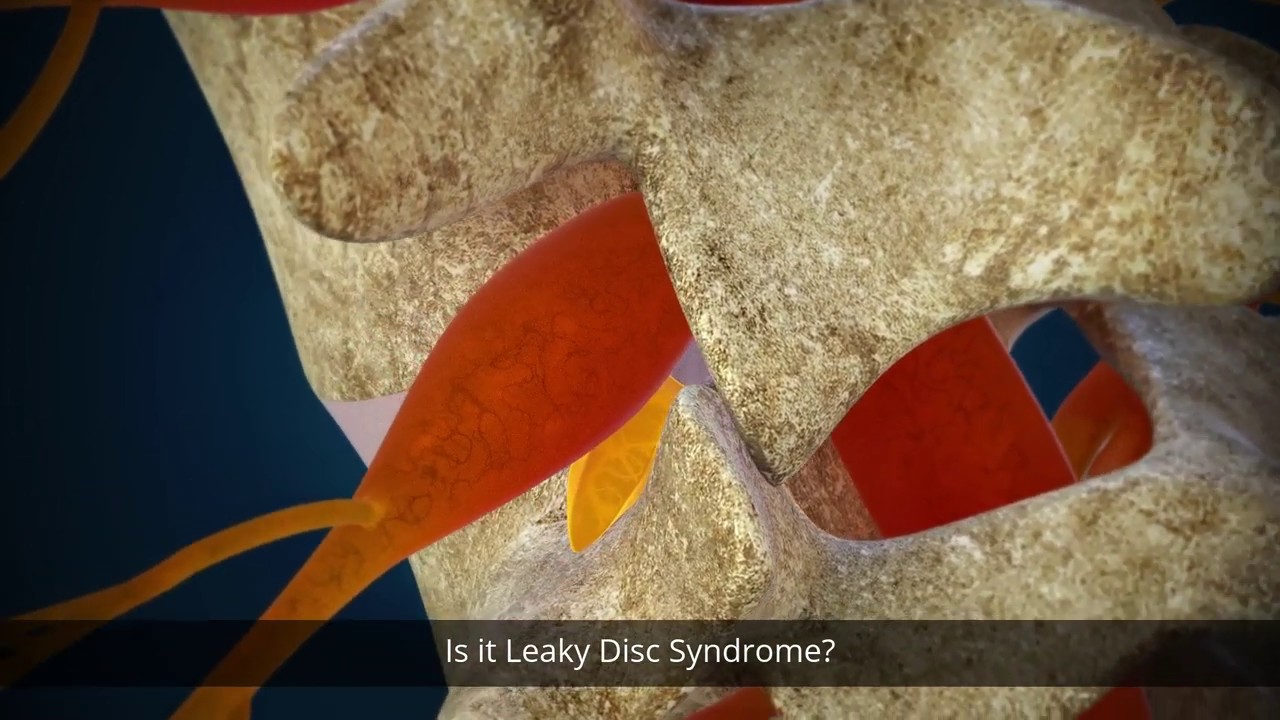
The Discseel Procedure is a revolutionary, minimally-invasive procedure to treat chronic back pain resulting from damaged or torn spinal discs. An orthobiologic is injected into the disc, which seals the tears and allows the body to begin healing. The Discseel Procedure is a true alternative to a spinal fusion.
Invented by Dr. Pauza, MD, the Discseel Procedure repairs damaged/torn spinal discs through the use of an orthobiologic. It is used to seal tears in your compromised discs. Spine surgeries, including spinal fusions and discectomies, can’t repair tears in the disc. The orthobiologic used in this procedure has proven to be an effective solution for tissue that cannot be sutured or ligatured together. Dr. Pauza recognized this, which led to the development of the Discseel® Procedure.
Surgeons have attempted to suture damaged or torn discs in the spine without success. The disc tears prevent regenerative medicine therapies, such as cell-based therapies and PRP to effectively repair the disc, as the biologic would seep out of the still-torn spinal disc. Over time, as your torn disc continues to leak, it can lose hydration and begin to degenerate. By using a particular orthobiologic, the Discseel Procedure not only treats the damaged and torn disc, but it also allows an individual’s body to replenish the lost cells with new cells, helping to restore the degenerated disc to a healthy state. Essentially, the goal of the Discseel Procedure is to seal the tears and instigate healthy growth of new disc tissue to replace the damaged or lost tissue in an effort to stop the disc from leaking and inflaming the nerves.
Dr. Tolman was carefully chosen by Dr. Kevin Pauza himself and trained to perform the Discseel® Procedure. Dr. Tolman is currently the only physician in the United States to perform the Discseel® Procedure on the thoracic spine.
Here's What to Expect
Consultation
During the in-person consultation you will speak directly to Dr. Tolman, who will perform the Discseel® Procedure. We want make sure you are confident about the procedure and that we can answer all your questions prior to the procedure.
Annulogram
The annulogram is a detailed process that will identify your pain source. It specifically determines if your normal or abnormal-looking discs are leaking. Anything missed by an MRI will show up on the annulogram.
Treatment
During the Discseel Procedure, Dr. Tolman will inject an orthobiologic into your damaged disc, which will seal the disc. The entire procedure is observed though live x-rays.
Recovery
After the Discseel® Procedure, most patients are walking within the first 24 hours. Immediately after the procedure, your damaged disc will begin healing, which is a process that will continue over the next 12 months.
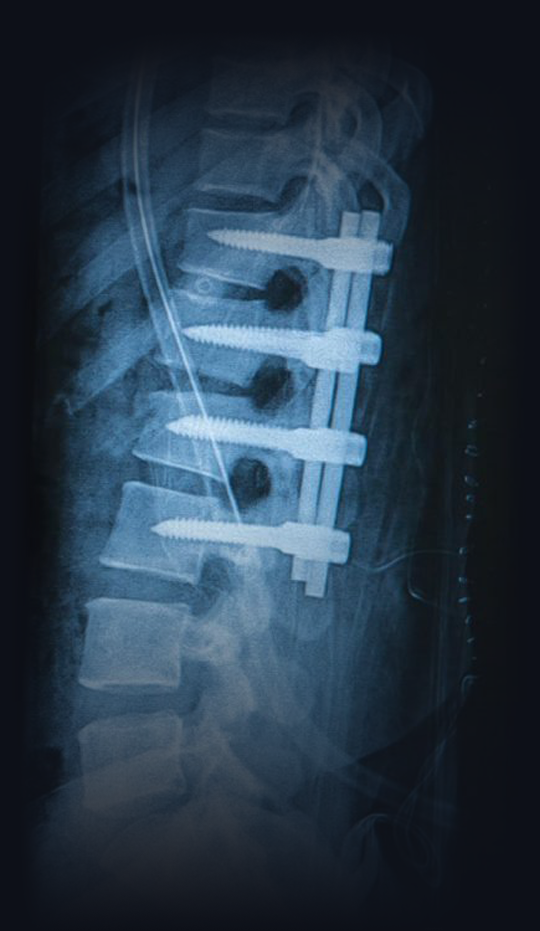
Spine treated with screws and pods that are used in spinal fusion surgery
The Discseel®
Procedure vs. Spinal Fusion
For decades, physicians have been recommending spinal fusions to patients, only for them to fail. These life-altering procedures have lead to some of the highest percentage of surgical failures and opioid dependency among surgical patients. So the real question is: why are doctors not recommending a procedure that has greater success? It’s simple. An average spinal fusion patient is worth between $250,000 and $750,000 in insurance payouts over the patient’s lifetime. It was the high failure that lead to the creation of the Discseel® Procedure. So how do the two procedures compare to eachother? Here are some facts:
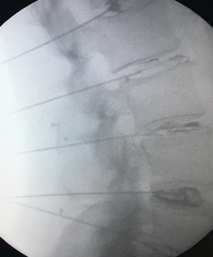
Spine treated with microscopic needles in the Discseel® Procedure
- In over 40% of patients that receive a spinal fusion, the fusion does not address the long term source of the pain
- At least 1/3 of all spinal fusion patients need a second or third fusion of their spine within 10 years
- The Discseel® Procedure has been successful in relieving back pain in 70% of patients that had a previous failed spinal procedure, such as a spinal fusion
- The Discseel® Procedure is a non-invasive outpatient procedure, whereas a spinal fusion is an invasive surgery that leaves scars, hardware and requires days of recovery in a hospital

Spine treated with screws and pods that are used in spinal fusion surgery



